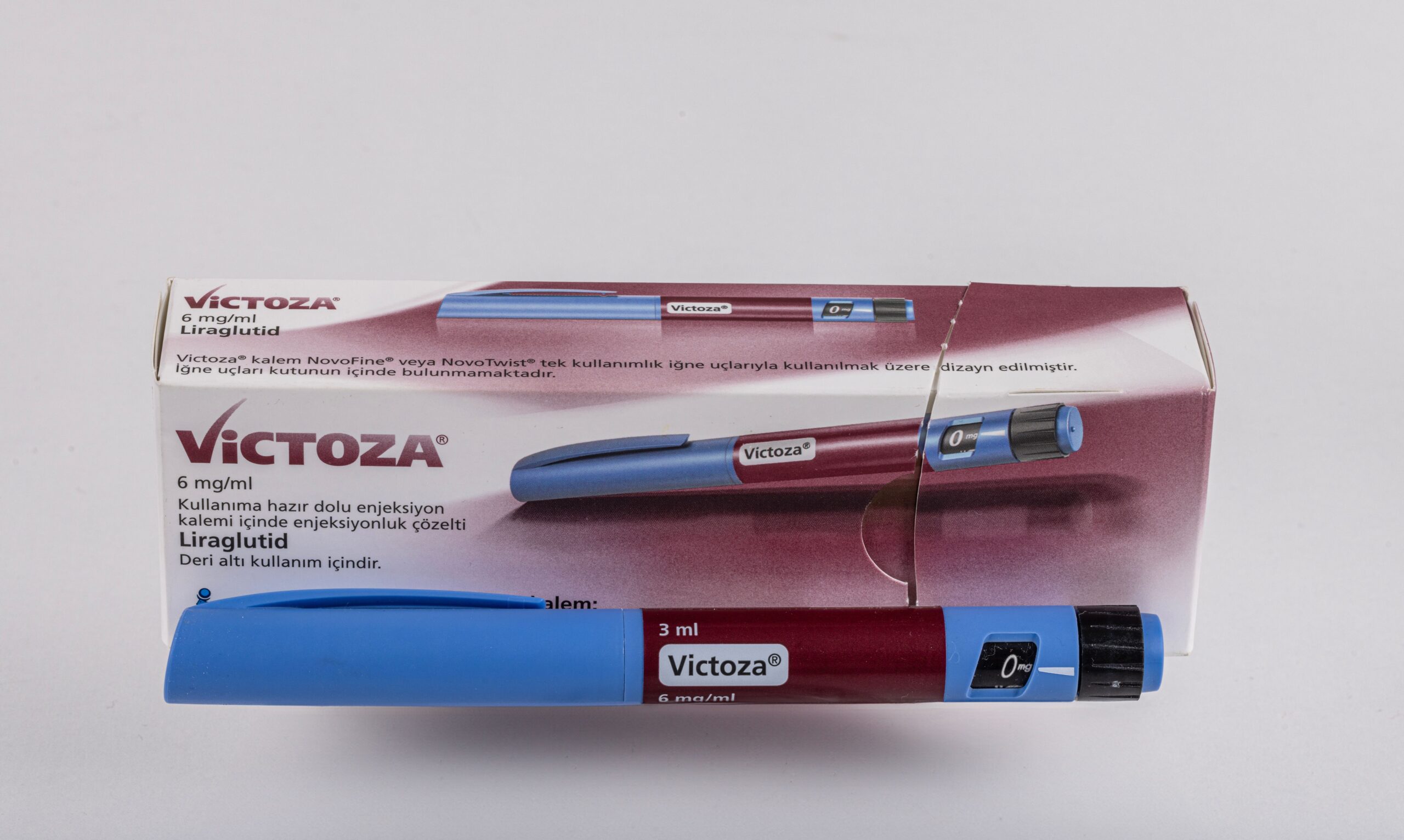
Victoza
$375.00
Victoza® (liraglutide) is a once-daily injectable GLP-1 receptor agonist medication for adults with type 2 diabetes. Available in a multi-dose pen with flexible dosing up to 1.8mg daily, it helps improve blood sugar control as an adjunct to diet and exercise, and may reduce cardiovascular risk in patients with established cardiovascular disease.
Daily GLP-1 Therapy for Comprehensive Diabetes Management
Victoza® (liraglutide) represents a well-established daily injectable option for type 2 diabetes management, offering patients and healthcare providers a flexible, titration-based approach to glycemic control. As a glucagon-like peptide-1 (GLP-1) receptor agonist, Victoza® works by mimicking the body’s natural incretin hormones to address multiple facets of diabetes pathophysiology. Administered once daily via a multi-dose pen, this medication provides glucose-dependent insulin secretion, suppresses inappropriate glucagon release, slows gastric emptying, and promotes satiety. With over a decade of clinical use and research, Victoza® has established itself as a cornerstone therapy for type 2 diabetes, particularly for patients who may benefit from daily administration or flexible dosing adjustments.
Flexible Dosing with Proven Cardiovascular Benefits
Victoza® follows a patient-specific titration schedule that begins at 0.6mg daily for one week to minimize gastrointestinal side effects, then increases to 1.2mg daily. Based on glycemic response and tolerability, the dose may be further increased to 1.8mg daily—the maximum recommended dose. This gradual, flexible approach allows for individualized treatment optimization. Importantly, Victoza® is the first GLP-1 receptor agonist to demonstrate cardiovascular outcome benefits in the landmark LEADER trial, showing significant reduction in major adverse cardiovascular events (MACE) in adults with type 2 diabetes and established cardiovascular disease. This dual benefit of glycemic control and cardiovascular risk reduction makes Victoza® a valuable option for patients with both metabolic and cardiovascular concerns.
Key Features of Victoza® Therapy:
-
Once-Daily Flexible Dosing: Administered at any time of day, independent of meals, offering convenience and adaptability to individual routines.
-
Proven Cardiovascular Risk Reduction: Demonstrated to significantly reduce the risk of major cardiovascular events in patients with type 2 diabetes and established cardiovascular disease.
-
Gradual Titration Schedule: Starts at 0.6mg daily for one week to improve tolerability, with stepwise increases to 1.2mg or 1.8mg based on individual needs.
-
Established Safety Profile: Over 10 years of real-world experience with well-characterized safety and efficacy data.
-
Weight-Neutral to Weight-Reducing Effects: Many patients experience weight loss or weight stabilization as a beneficial effect.
Clinical Protocol & Important Safety Information:
-
Dosage Schedule: Begin at 0.6mg daily for one week to reduce gastrointestinal symptoms, then increase to 1.2mg daily. If needed for additional glycemic control, may increase to 1.8mg daily after at least one week at 1.2mg.
-
Administration: Injected subcutaneously once daily at any time, independent of meals. Use the Victoza® multi-dose pen with disposable needles.
-
Injection Sites: Abdomen, thigh, or upper arm. Rotate injection sites daily to reduce the risk of lipodystrophy.
-
Treatment Duration: Intended for long-term use as part of diabetes management. Patients should be periodically reassessed for continued need and effectiveness.
-
Common Side Effects: Nausea, diarrhea, headache, vomiting, constipation, and indigestion. Gastrointestinal side effects are most common during initiation and dose escalation and typically diminish over time.
-
Important Safety Information:
-
Boxed Warning: Risk of thyroid C-cell tumors. Contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
-
Pancreatitis: Discontinue if pancreatitis is suspected. Do not restart if pancreatitis is confirmed.
-
Hypoglycemia: Risk increases when used with insulin or insulin secretagogues. May require dose adjustment of these concomitant medications.
-
Renal Impairment: Use with caution in patients with renal impairment; monitor renal function in patients reporting severe gastrointestinal reactions.
-
Cardiovascular Indication: To reduce the risk of major adverse cardiovascular events in adults with type 2 diabetes mellitus and established cardiovascular disease.
-
-
Medical Supervision: Requires prescription and ongoing monitoring by healthcare provider. Regular follow-up is essential to assess glycemic control, tolerability, and need for continued therapy.
Be the first to review “Victoza” Cancel reply
Related products
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes




Reviews
There are no reviews yet.