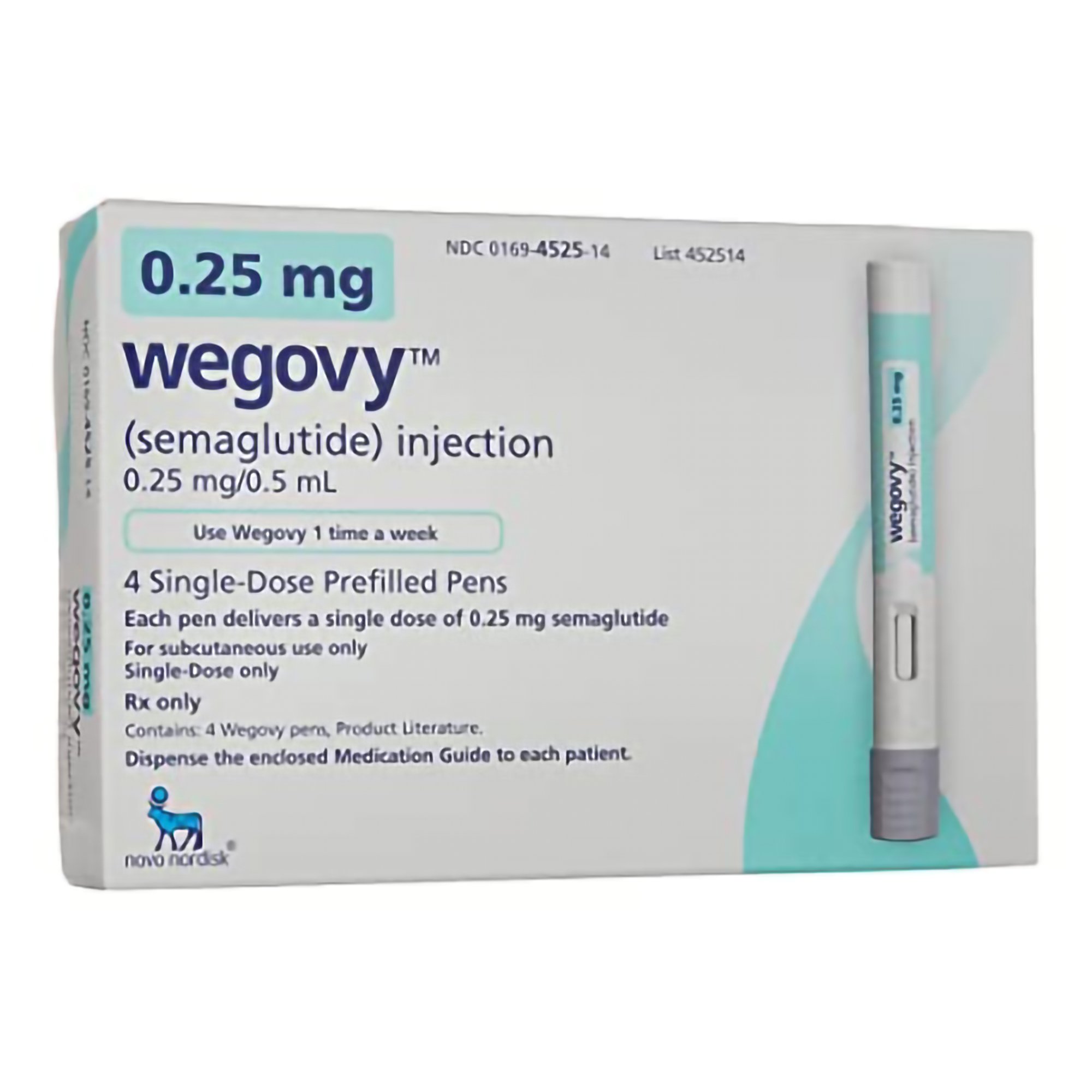
Wegovy 0.25mg
$180.00
The Wegovy® 0.25mg dose is the starting dose for adults with obesity or overweight with weight-related medical problems, beginning a structured weight management program. This once-weekly GLP-1 receptor agonist injection helps your body gradually adjust to therapy while initiating appetite regulation under medical supervision.
The Foundational Step in Medical Weight Management
The Wegovy® 0.25mg dose represents the crucial first step in a medically-supervised weight management program for adults with obesity (BMI ≥30) or overweight (BMI ≥27) with at least one weight-related comorbidity. As the initiation dose of semaglutide specifically formulated for chronic weight management, this 0.25mg weekly injection is designed to gently introduce the body to GLP-1 receptor agonist therapy while minimizing potential gastrointestinal side effects. Delivered through Novo Nordisk’s proprietary pre-filled pen, this starter dose establishes the foundation for sustainable weight reduction by beginning to regulate appetite and food intake through gradual, tolerable engagement of the body’s natural satiety pathways.
Gentle Introduction to Appetite Regulation
The 0.25mg once-weekly dose serves as a four-week initiation period where the medication begins to activate GLP-1 receptors involved in appetite regulation and gastric emptying. At this starting strength, Wegovy® initiates a mild slowing of gastric transit and starts to enhance feelings of fullness after meals while influencing central nervous system pathways that control hunger and cravings. This gradual introduction is clinically proven to significantly improve gastrointestinal tolerability, with studies showing reduced incidence of nausea and other digestive side effects compared to starting at higher doses. Patients typically notice early effects on portion control and reduced food cravings during these first four weeks, establishing positive behavioral patterns that support long-term weight management success.
Key Features of the 0.25mg Starter Dose:
-
Tolerability-Focused Initiation: Specifically formulated as a starting dose to minimize common side effects like nausea, vomiting, and diarrhea while your body adjusts to GLP-1 receptor activation.
-
Structured Treatment Pathway: Follows evidence-based clinical guidelines recommending four weeks at 0.25mg before dose escalation, ensuring safe progression in a medically-supervised weight management program.
-
Early Appetite Regulation: Begins activating GLP-1 receptors to enhance satiety signaling, reduce hunger, and help establish healthier eating patterns from treatment inception.
-
Same Delivery System: Uses the identical, user-friendly Wegovy® pre-filled pen device as higher doses, allowing patients to become comfortable with the once-weekly injection routine.
-
Behavioral Foundation: Supports the development of sustainable lifestyle habits during the initial treatment phase, complementing the comprehensive weight management program that includes reduced-calorie diet and increased physical activity.
Usage Protocol & Important Safety Information:
-
Dosage Regimen: One subcutaneous injection of 0.25mg once weekly for the first four weeks of treatment. Inject on the same day each week, with or without meals.
-
Administration Sites: May be injected into the abdomen, thigh, or upper arm. Rotate injection sites weekly to reduce the risk of lipodystrophy.
-
Device Features: The Wegovy® 0.25mg dose comes in a pre-filled, single-dose pen. The pen features a hidden needle, simple dose selection, and an easy-to-press injection button.
-
Titration Schedule: After four weeks at 0.25mg, the dose should be increased to 0.5mg once weekly under healthcare provider guidance. Do not exceed the 0.25mg dose beyond the initial four weeks unless specifically directed by your physician.
-
Common Side Effects at Initiation: Nausea, diarrhea, vomiting, constipation, abdominal pain, headache, and fatigue may occur but are typically mild to moderate and diminish over time as the body adjusts.
-
Boxed Warning: Risk of thyroid C-cell tumors. Wegovy® is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
-
Important Contraindications: Not for patients with a personal or family history of MTC or MEN 2. Not for use during pregnancy. Use with caution in patients with history of pancreatitis, gallbladder disease, or severe gastrointestinal disease.
-
Medical Supervision Required: This medication requires a prescription and ongoing monitoring by a healthcare professional. Regular follow-up is essential to assess tolerability, progress, and determine appropriate dose escalation.
Be the first to review “Wegovy 0.25mg” Cancel reply
Related products
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes







Reviews
There are no reviews yet.